 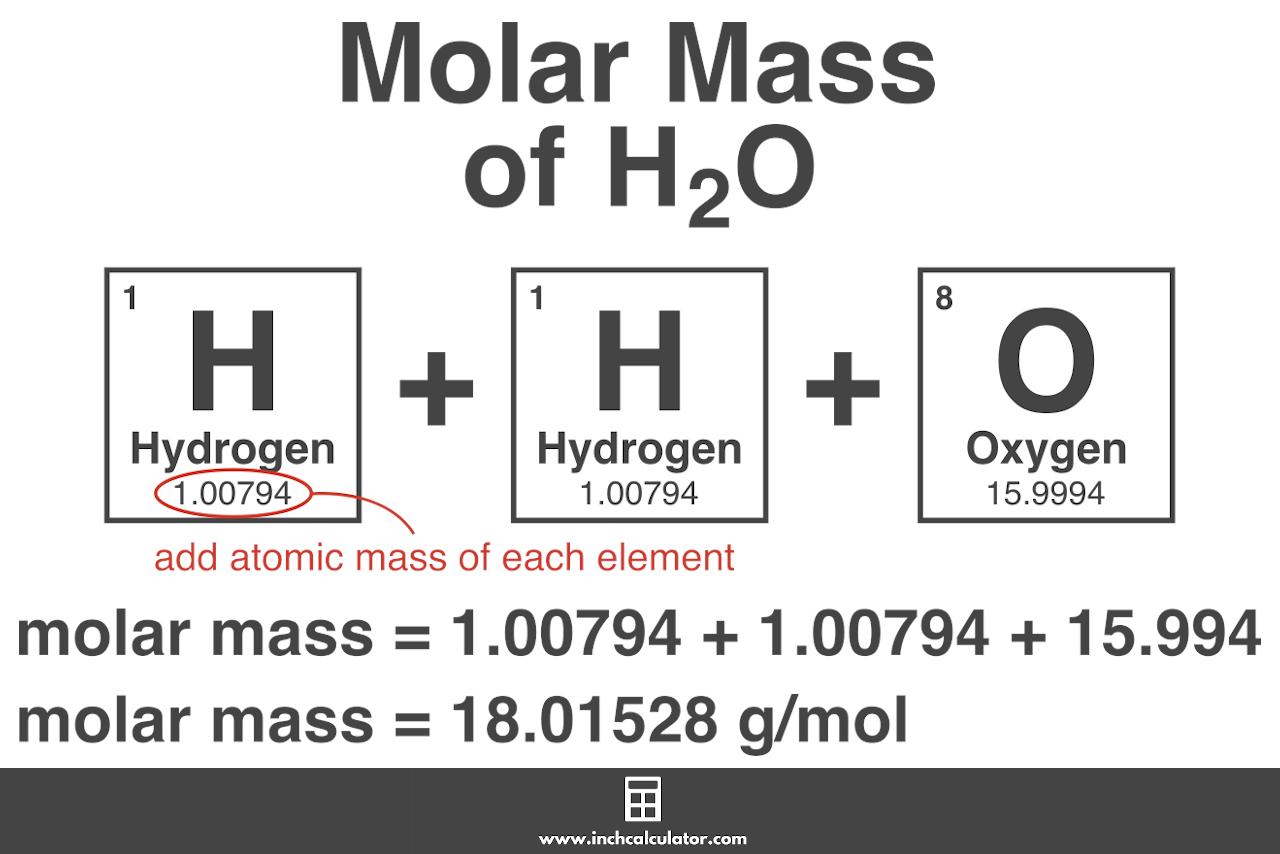
So how good is our atomic radius? Standard tables give the atomic radius of strontium is in the range 192-220 pm. molar mass of carbon-12: Numerical value: 11.9 x 10-3 kg mol-1: Standard uncertainty: 0.0 x 10-3 kg mol-1: Relative standard uncertainty: 3. molar mass of carbon-12: Numerical value: 11.9 x 10-3 kg mol-1: Standard uncertainty: 0.0 x 10-3 kg mol-1: Relative standard uncertainty: 3.0 x 10-10: Concise form 11. Since 3 3=27 and 4 3 = 64, you know that the cube root of 55 will be between 3 and 4, so the cube root should be a bit less than 4 × 10 –8. Element: Boron Symbol: B Atomic Mass: 10. Element: Lanthanum Symbol: La Atomic Mass: 138.9055 of Atoms: 1 Mass Percent: 47.311. The mass number is nothing but the sum of the number of protons and number of neutrons present in the nucleus of the atom.\), for example. Calculate the molar mass of CArBON BLaCK in grams per mole or search for a chemical formula or substance. Molecular weight is also called mass number. Therefore, the answer is – option (c) – Gram molecular mass of carbon is 12g It has an atomic mass of 12 atomic mass units or 12g. The molar mass of any substance is numerically equivalent to its atomic or formula weight in amu. It is a p-block element, present in Group 14 and 2nd period. To start, determine the number of moles of carbon per 1 mole of C13H18O2. \Ĭarbon is an element with atomic number 6. The mass of a water molecule, H2O, can be referred to as a molecular mass. Molar mass of an element and atomic mass of an element have the same value. The molar mass is defined as the mass of a given substance divided by the amount of a substance and is expressed in g/mol. It is due to the fact that the atoms of different elements contain different numbers of protons, neutrons, and electrons, and therefore they have different masses. Solution Molecules of this compound are composed of 13 carbon atoms, 18 hydrogen atoms, and 2 oxygen atoms. All elements have different molar masses. It should be noted that each element has its own unique molar mass. moles C13H18O2 to grams Molecular weight calculation: 12.010713 1.0079418 15.99942 Percent composition by element Element: Hydrogen Symbol: H Atomic Mass: 1.00794 of Atoms: 18 Mass Percent: 8.795 Element: Carbon Symbol: C Atomic Mass: 12.0107 of Atoms: 13 Mass Percent: 75.692 Element: Oxygen Symbol: O Atomic Mass: 15. 
LEWIS DOT STRUCTURE OF MOLECULE NH C,H, CH CO. “The mass in grams of one mole of substance is called molar mass”. What is the molecular formula of this compound VALENCE TOTAL OF ELECTRONS FROM VALENCE EACH ATOM ELECTRONS FORMULA PO. Refer to the periodic table to solve this question. Get control of 2022 Track your food intake, exercise, sleep and meditation for free. We can then use the calculated molar mass to convert between mass and number of moles of the substance. Molar Mass, Molecular Weight and Elemental Composition Calculator. As shown in this video, we can obtain a substance's molar mass by summing the molar masses of its component atoms. On the basis of above data, the average atomic mass of carbon will be. About Transcript The molar mass of a substance is the mass in grams of 1 mole of the substance. Atomic number refers to the number of electrons present in the element. Isotope Relative abundance() Atomic mass(amu) 12 C 98.892 12 13 C 1.108 13.00335 14. Hint: Do not confuse atomic mass with atomic number. The molar mass is defined as the mass of a given substance divided by the amount of a substance and is expressed in g/mol. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
